#ethylenediaminetetraphosphonate
Text
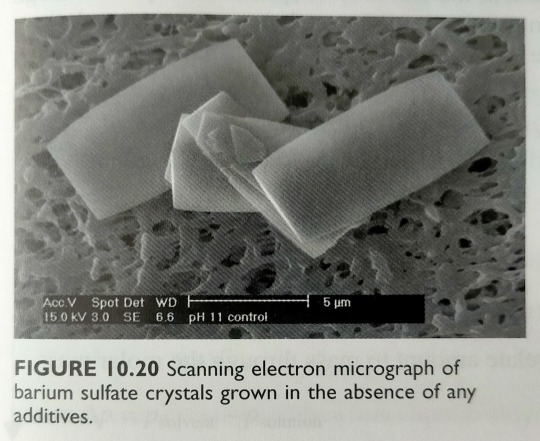
Experimentally, we usually observe changes in the crystal shape – a clear sign that the additive is binding to the crystal surface and changing the relative rates of growth of different crystal faces (figures 10.20 and 10.21).

"Chemistry" 2e - Blackman, A., Bottle, S., Schmid, S., Mocerino, M., Wille, U.
#book quotes#chemistry#nonfiction#textbook#experiments#observation#crystals#microscopy#scanning electron micrograph#barium sulfate#ethylenediaminetetraphosphonate#anion
0 notes
Text
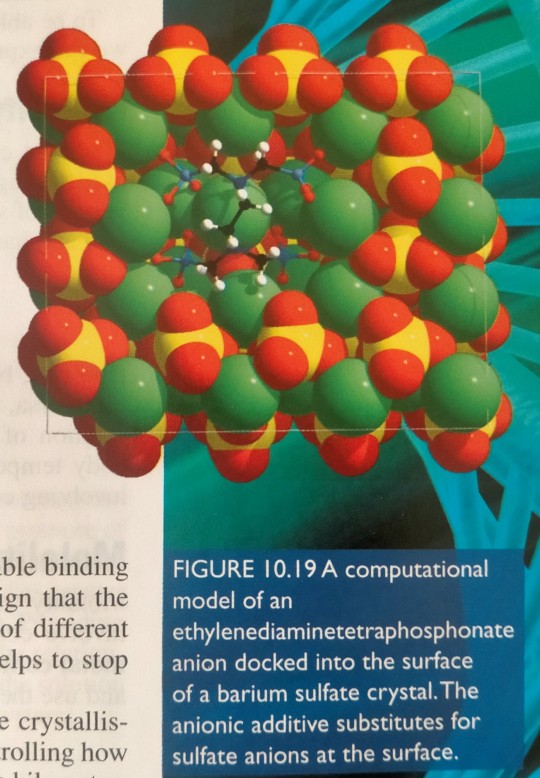
For example, additives such as the ethylenediaminetetraphosphonate anion can substitute for sulfate anions at the surface of a barium sulfate crystal; the system can be modelled in the computer (figure 10.19) to determine the most favourable binding sites.
"Chemistry" 2e - Blackman, A., Bottle, S., Schmid, S., Mocerino, M., Wille, U.
#book quotes#chemistry#nonfiction#textbook#additives#ethylenediaminetetraphosphonate#anion#ions#sulfate#substitution#barium sulfate#computer modeling
0 notes