#eosinophilic disorders
Text

#eosinophilic gastritis#chronic illness#eosinophilic disorders#chronic illness meme#rare disease#EoG#eosinophilic disease#eosinophilic gastrointestinal disorders#eosinophilic gastrointestinal disease#monoclonal antibody#immune system disorder#immune system#pharmacology#biotech#biotechindustry#medication#medicine#rare disorders#il-5#fasenra#interleukin-5#benralizumab
7 notes
·
View notes
Note
only if you’re comfy ofc — but would you explain what it was like to get diagnosed with eoe? I have a different eos disorder, but until i asked them to test me for my illness, my doctors consistently ignored my high eos counts and blamed it on allergies… lowkey just wanna know if all doctors are that shitty or if it’s just mine lol
i don’t mind at all !!!
we actually accidentally found my eoe
i’d been tested for allergies multiple times bc i get allergy-like symptoms (don’t know why exactly bc no mcas or allergy presentations other than eoe) but every test has always been negative and idk if we ever looked at my eosinophils in my blood
i was experiencing general gi symptoms that we assumed were gastroparesis but my doctor wanted me to have an upper endoscopy and colonoscopy to rule out anything else !
during that endoscopy/colonoscopy, everything was fine except my doc found the eosinophils and biopsied to confirm. he immediately gave me the diagnosis day of.
my family and i had never heard of eoe at all and my doctor was not looking for it so it came as a surprise but it was a good catch as i’ve most likely had this for years (if not my whole life) and now we can treat it before i get permanent strictures in my throat !
#actually disabled#disability#chronic illness#disabled#chronically ill#cripple punk#cripplepunk#spoonie#cpunk#physically disabled#eoe#eosinophilic esophagitis#eosinophils#eosinophilic disorder#kaz’s asks
10 notes
·
View notes
Link
Although gluten is believed to be a potential trigger for symptoms, other components of wheat may also be triggers, including fructans, alpha-amylase trypsin inhibitors, and wheat germ agglutinins. Several primary gastrointestinal disorders are associated with an increase in inflammatory cells including eosinophils.
Diet seems to be an important driver of disease pathogenesis in eosinophilic gastroenteritis, with elimination and elemental diets showing promise in management, with further robust trials required. Small intestinal bacterial overgrowth is an example of microbial dysbiosis, with renewed interest in diet being postulated to cause an adaptive change of the microbes colonizing the small intestine. However, the diagnosis of small intestinal bacterial overgrowth is limited by a lack of sensitive and specific tests, with significant knowledge gaps in relation to therapeutic measures to manage and cure small intestinal bacterial overgrowth. Currently, antimicrobials are the established management option. There have been significant clinical advances in dietary interventions related to the small bowel, but this area is currently a novel and advancing field for both patients and clinicians.
#SIBO#SIFO#Candida#Candidasis#small bowl disorders#gluten#eosinophilic gastroenteritis#elimination diet
0 notes
Text

Eosinophilic disorders include eosinophilic cystitis, eosinophilic fasciitis, eosinophilic pneumonia, eosinophilic gastrointestinal disorders, eosinophilic granulomatosis with polyangiitis and eosinophilia.
0 notes
Text
Thinking of people with dietary restrictions this Disability Pride Month. Shoutout to:
People with dietary restrictions due to allergies
People with dietary restrictions due to related disorders, like eosinophilic esophagitis or MCAS
People with dietary restrictions due to autoimmune disorders
People with dietary restrictions due to sensory issues and/or ARFID
People with dietary restrictions due to plain old food intolerances
People with any combination of the above
People whose allergens/intolerances/etc are never labeled properly on ingredient lists
People constantly seeing "X ingredient free!" labels that just straight-up lie
People whose allergens/intolerances/etc are additives used in fucking everything
People who can't figure out what their trigger foods are, because their prime suspects are inescapably included in fucking everything
People who have such a hard time avoiding every trigger that we're just chronically ill now, apparently
People whose safe foods are impossible to find in stores consistently
People who had safe store-bought foods until they fucking changed the recipe
People who can't travel, because they can't guarantee access to their safe foods while away from home
People whose social life suffers, because they can't go out to eat at a restaurant or get coffee with friends
People with social anxiety about standing out as the only person with a pre-packed lunch and getting questioned about it
People who fell in love with cooking and baking for themselves, but started out with it because it was the only way the could get their needs met
People who can't cook or bake for themselves because of the time commitment, or because of any another disability they have
People who still just struggle with cooking, for any reason
People who miss that one food so so much
People who can never get a straight answer from their workplace about what food is going to be provided for that special lunch, or even if it will be catered versus going out to a restaurant
People whose dietary restrictions are ones that no one has heard of, or is willing to believe
People who would be vegetarian or vegan were it not for their dietary restrictions, but need animal products to live
People who are constantly left out of conversations on the environmentalism and ethics of food consumption because of this
People living places with dogshit healthcare systems, who can't afford to pay for an EpiPen or other life-saving medication
People with social trauma from being forced to sit alone at the "allergy only" table in elementary school lunch
People sick and tired of how all the media discussion and advocacy for people with dietary restrictions focuses on elementary schoolers
People with long term health complications from prior exposure to their trigger foods
People with trauma from medical episodes or the long term health complications
People with trauma from medical institutions not taking their dietary restrictions seriously
People who, despite everything, only hear their dietary restrictions mentioned when they're the butt of a joke
People who, despite everything, get told they're "just complaining" or "having a first world problem"
People who, despite everything, get told they don't count as having a disability
I see you. I'm with you. And you are under no obligation to be quiet about what you go through. We deal with a lot. But we're here for each other. And we are allowed to be visible and angry.
100 notes
·
View notes
Text
The CDC has quietly changed who should AVOID the MMR vaccine.
https://www.cdc.gov/vaccines/vpd/mmr/public/index.html
They now state that ANYONE that “Has a parent, brother or sister with a history of immune system problems” should AVOID THE MMR VACCINE!
What exactly is an 'immune system problem?" Every autoimmune disorder.
* Achalasia
* Addison’s disease
* Adult Still's disease
* Agammaglobulinemia
* Alopecia areata
* Amyloidosis
* Amyotrophic lateral sclerosis (Lou Gehrigs)
* Ankylosing spondylitis
* Anti-GBM/Anti-TBM nephritis
* Antiphospholipid syndrome
* Autoimmune angioedema
* Autoimmune dysautonomia
* Autoimmune encephalomyelitis
* Autoimmune hepatitis
* Autoimmune inner ear disease (AIED)
* Autoimmune myocarditis
* Autoimmune oophoritis
* Autoimmune orchitis
* Autoimmune pancreatitis
* Autoimmune retinopathy
* Autoimmune urticaria
* Axonal & neuronal neuropathy (AMAN)
* Baló disease
* Behcet’s disease
* Benign mucosal pemphigoid
* Bullous pemphigoid
* Castleman disease (CD)
* Celiac disease
* Chagas disease
* Chronic inflammatory demyelinating polyneuropathy (CIDP)
* Chronic recurrent multifocal osteomyelitis (CRMO)
* Churg-Strauss Syndrome (CSS) or Eosinophilic Granulomatosis (EGPA)
* Cicatricial pemphigoid
* Cogan’s syndrome
* Cold agglutinin disease
* Congenital heart block
* Coxsackie myocarditis
* CREST syndrome
* Crohn’s disease
* Dermatitis herpetiformis
* Dermatomyositis
* Devic’s disease (neuromyelitis optica)
* Discoid lupus
* Dressler’s syndrome
* Endometriosis
* Eosinophilic esophagitis (EoE)
* Eosinophilic fasciitis
* Erythema nodosum
* Essential mixed cryoglobulinemia
* Evans syndrome
* Fibromyalgia
* Fibrosing alveolitis
* Giant cell arteritis (temporal arteritis)
* Giant cell myocarditis
* Glomerulonephritis
* Goodpasture’s syndrome
* Granulomatosis with Polyangiitis
* Graves’ disease
* Guillain-Barre syndrome
* Hashimoto’s thyroiditis
* Hemolytic anemia
* Henoch-Schonlein purpura (HSP)
* Herpes gestationis or pemphigoid gestationis (PG)
* Hidradenitis Suppurativa (HS) (Acne Inversa)
* Hypogammalglobulinemia
* IgA Nephropathy
* IgG4-related sclerosing disease
* Immune thrombocytopenic purpura (ITP)
* Inclusion body myositis (IBM)
* Interstitial cystitis (IC)
* Juvenile arthritis
* Juvenile diabetes (Type 1 diabetes)
* Juvenile myositis (JM)
* Kawasaki disease
* Lambert-Eaton syndrome
* Leukocytoclastic vasculitis
* Lichen planus
* Lichen sclerosus
* Ligneous conjunctivitis
* Linear IgA disease (LAD)
* Lupus
* Lyme disease chronic
* Meniere’s disease
* Microscopic polyangiitis (MPA)
* Mixed connective tissue disease (MCTD)
* Mooren’s ulcer
* Mucha-Habermann disease
* Multifocal Motor Neuropathy (MMN) or MMNCB
* Multiple sclerosis
* Myasthenia gravis
* Myositis
* Narcolepsy
* Neonatal Lupus
* Neuromyelitis optica
* Neutropenia
* Ocular cicatricial pemphigoid
* Optic neuritis
* Palindromic rheumatism (PR)
* PANDAS
* Parkinson's disease
* Paraneoplastic cerebellar degeneration (PCD)
* Paroxysmal nocturnal hemoglobinuria (PNH)
* Parry Romberg syndrome
* Pars planitis (peripheral uveitis)
* Parsonage-Turner syndrome
* Pemphigus
* Peripheral neuropathy
* Perivenous encephalomyelitis
* Pernicious anemia (PA)
* POEMS syndrome
* Polyarteritis nodosa
* Polyglandular syndromes type I, II, III
* Polymyalgia rheumatica
* Polymyositis
* Postmyocardial infarction syndrome
* Postpericardiotomy syndrome
* Primary biliary cirrhosis
* Primary sclerosing cholangitis
* Progesterone dermatitis
* Psoriasis
* Psoriatic arthritis
* Pure red cell aplasia (PRCA)
* Pyoderma gangrenosum
* Raynaud’s phenomenon
* Reactive Arthritis
* Reflex sympathetic dystrophy
* Relapsing polychondritis
* Restless legs syndrome (RLS)
* Retroperitoneal fibrosis
* Rheumatic fever
* Rheumatoid arthritis
* Sarcoidosis
* Schmidt syndrome
* Scleritis
* Scleroderma
* Sjögren’s syndrome
* Sperm & testicular autoimmunity
* Stiff person syndrome (SPS)
* Subacute bacterial endocarditis (SBE)
* Susac’s syndrome
* Sympathetic ophthalmia (SO)
* Takayasu’s arteritis
* Temporal arteritis/Giant cell arteritis
* Thrombocytopenic purpura (TTP)
* Tolosa-Hunt syndrome (THS)
* Transverse myelitis
* Type 1 diabetes
* Ulcerative colitis (UC)
* Undifferentiated connective tissue disease (UCTD)
* Uveitis
* Vasculitis
* Vitiligo
* Vogt-Koyanagi-Harada Disease
Wonder how many doctors are paying attention?
~shared from Jodi Wilson
28 notes
·
View notes
Text
Aplastic Anemia | Causes, Symptoms, Diagnosis and Treatments
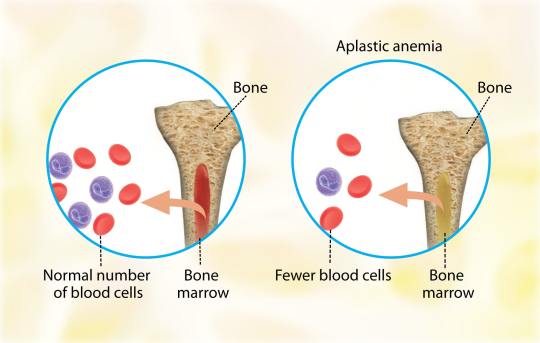
Aplastic anemia (AA) is the syndrome of chronic primary hematopoietic failure caused by injury, which results in decreased or absent hematopoietic precursors in the bone marrow and accompanying pancytopenia.
Aplastic anemia is classified as either moderate, severe (SAA), or very severe (vSAA).
Three primary mechanisms can result in the development of AA: –
1. Direct injury.
2. Immune-mediated.
3. Inherited or acquired bone marrow failure.
Usually, AA is idiopathic, however, it can be attributable to: –
- Radiation.
- Toxic chemicals (like Benzene, solvents, and glue vapors).
- Cytotoxic drugs (chloramphenicol, gold).
- Immune-related disorders (Eosinophilic fasciitis, SLE, Graft versus host disease).
- Thymoma.
- Viral infections (Epstein-Virus Infection, Parvovirus B19, Human immunodeficiency virus (HIV), and Hepatitis virus).
- Anorexia nervosa and paroxysmal nocturnal hemoglobinuria (PNH).
Patients typically present with non-specific symptoms caused by associated cytopenia, such as: –
- Low energy levels, pallor, and headaches with anemia.
- Mucosal bleeding.
- Bruising/petechiae.
- Menorrhagia with thrombocytopenia.
- Fever with or without evidence of infection with neutropenia.
The most common complications of aplastic anemia are bleeding, infections, and transformation to lymphoproliferative disorders.
Aplastic anemia has the following diagnostic criteria: –
The presence of bone marrow hypocellularity and two or more cytopenias (reticulopodia less than 1% or less than 40,000/microliter, neutropenia less than 500/microliter, or thrombocytopenia less than 20,000/microliter).
The bone marrow cellularity in moderate disease is less than 30%.
The severe disease has less than 25% cellularity or less than 50% cellularity with fewer than 30% hematopoietic cells.
Very severe meets the severe criteria in addition to neutropenia less than 200/µL.
Aplastic anemia treatment focuses on the underlying cause.
If possible, remove the offending agent(s).
Treatment is dependent on the patient’s age, disease severity, donor availability, and performance status when there is no distinctive reversible cause.
Young patients (less than 50 years old) with severe disease who are in good health should receive an allogeneic hematopoietic cell transplant (HCT) before starting immunosuppressive therapy.
Older patients (50 years and older) in good health, as well as young patients without an HCT donor, are given full-dose immunosuppressive therapy using: –
- Eltrombopag.
- Horse/rabbit anti-thymocyte globulin (ATG).
- Cyclosporine A.
- Prednisone.
Read more at: https://medicaregate.com/aplastic-anemia-causes-symptoms-diagnosis-and-treatments/
2 notes
·
View notes
Text
—About Me— (this is the fun part!)
Name: Annabelle Rae
I also go by: Anna, Annie, Jasper
Part of the LGBTQIA (non-binary & genderfluid)
Sexual orientation: Asexual, bisexual
Pronouns: she/her & they/them
Additional Info:
• Autistic
• ADHD
• Auditory processing disorder
Disabling conditions I live with:
• GERD/esophageal motility issues
• Intracranial pressure
• Chronic pain, widespread
• Possible HSD
• Eosinophilic asthma
• Tracheomalacia (the fancy medical term for floppy trachea)
• Chronic migraines
• Severe mental illnesses: BPD, binge eating disorder, bipolar 1 with psychotic features, CPTSD, OCD, GAD, traits of agoraphobia, substance use disorder (sober so far!)
DOC (drugs of choice): meth, crack cocaine, alcohol, cocaine (in powder form)
Massive content/trigger warning: SA, bullying, abuse, suicidal ideation, ED thoughts, self harm, drug abuse
DO NOT REPORT. Unfollow or block. Love y’all 💜
2 notes
·
View notes
Text

#eosinophilic gastritis#eosinophilic disorders#rare disease#eosinophilic disease#eosinophilic gastrointestinal disorders#eosinophilic gastrointestinal disease#chronic illness#chronic illness meme#rare disorders#EoG#food#food intolerances#food allergies#elimination diet
6 notes
·
View notes
Text
Okay I’m moving to mobile. Gonna be out all day doing deliveries. 😩 the grind never stops.
I sent an email to my boss today before I left asking if working part time could be an option. I doubt it will be considering someone has to do my work whenever I am out… but it’s the only way I can think of help me get back on Medicaid. That or lowering my salary..
My doctor also wants me to get labs done tomorrow so I’m gonna do that in the morning. There’s real concern for my eyes, he thinks it’s possible my blood disorder could also be causing my eyesight to go bad. So he wants to check my eosinophils levels and see if the nucala is actually working.
I’ve got a lot of shit going on, and I’m trying my best to not fall into a ditch of self loathing and suicide ideation but my dudessss it’s hard. 
#ooc. // 𝐛𝐞 𝐬𝐭𝐢𝐥𝐥 𝐝𝐢𝐚 𝐬𝐩𝐞𝐚𝐤𝐬#I don’t have a therapist anymore#no one to talk to about it#life is hard rn#and I can feel how much it’s affecting me cuz I really just wanna. curl up and do nothing#and this time I don’t have my therapist to help me through it#life just feels very hopeless right now but I promise im trying
0 notes
Text
Blood cell

The blood cells serves as a comprehensive guide to understanding the intricacies of hematopoiesis, the process through which various blood cell types are formed and function in the human body. This detailed discussion aims to unravel the key aspects presented in the article, delving into the structure, functions, and disorders associated with;
Red blood cells (erythrocytes),
2.White blood cells (leukocytes), and
platelets (thrombocytes).
Blood Cell Types and Composition
At the core of the circulatory system lie three major types of blood cells: red blood cells, white blood cells, and platelets. These cellular components collectively contribute to 45% of blood tissue by volume, with the remaining 55% being plasma. This delicate balance underscores the dynamic nature of blood, serving as a conduit for various vital functions within the body.
1.Red Blood Cells (Erythrocytes)
The discussion commences with a focus on red blood cells, the primary carriers of oxygen in the bloodstream. Erythrocytes, characterized by their unique biconcave shape and lack of a nucleus, play a crucial role in gas exchange facilitated by the iron-containing protein hemoglobin. The intricate details of erythropoiesis, the process of RBC formation in the red bone marrow, offer a glimpse into the remarkable physiological mechanisms that ensure a constant supply of oxygen carriers. The staggering production rate of 2.4 million RBCs per second in adults highlights the body’s continuous demand for these essential cells. The information regarding the lifespan of RBCs (100–120 days) and their subsequent removal by the spleen adds another layer to our understanding of the life cycle of these vital cells. The absence of a nucleus in mature red blood cells, a unique characteristic among human cells, is highlighted. The pathological conditions of anemia and polycythemia are thoroughly explored, shedding light on the consequences of an imbalance in red blood cell count. Additionally, the erythrocyte sedimentation rate (ESR) provides valuable insights into the diagnostic tools used in assessing the health of red blood cells.
2.White Blood Cells (Leukocytes)
The immune system, our body’s defense mechanism, relies on white blood cells to combat infectious diseases and foreign materials. These leukocytes, originating from multipotent cells in the bone marrow, are categorized into granulocytes (basophils, eosinophils, neutrophils, mast cells) and agranulocytes (lymphocytes and monocytes). The discussion delves into the roles these distinct white blood cell types play in the human immune system, offering a nuanced understanding of their functions. The conditions of leukopenia and leukocytosis, indicating low and high white blood cell counts, respectively, are explored, emphasizing the diagnostic significance of monitoring these counts. The increased white blood cell count during infections and its association with hematological cancers underscore the pivotal role leukocytes play in our overall health.
3.Platelets (Thrombocytes)
The section on platelets elucidates their role in hemostasis, the process of preventing and stopping bleeding. These small, irregularly shaped cell fragments, derived from megakaryocytes, circulate in the blood and are essential for the formation of blood clots. The average lifespan of platelets, a mere 5 to 9 days, emphasizes the continuous production required for maintaining hemostatic balance. The normal range of platelet counts and the potential consequences of low or high platelet numbers provide valuable insights into the delicate equilibrium necessary for preventing excessive bleeding or the formation of thrombosis. Thrombocytopathy, a broad term encompassing disorders related to platelets, is discussed, including thrombocytopenia, thrombasthenia, and thrombocytosis. The intricate relationship between platelets and growth factors, as well as their role in wound healing, showcases the multifaceted contributions of these small but crucial cellular fragments.
Complete Blood Count (CBC): The article introduces the Complete Blood Count (CBC) as a vital diagnostic tool providing a comprehensive analysis of blood cell composition. The historical transition from manual counting to automated analyzers reflects the evolving landscape of medical technology, enhancing the efficiency and accuracy of blood cell analysis. The significance of CBC in offering an overview of a patient’s general health status is underscored, emphasizing its widespread use in medical diagnostics.
Historical Discoveries.
The historical perspective woven into the article traces the evolution of our understanding of blood cells. From Jan Swammerdam’s pioneering observation of red blood cells in 1658 to Paul Ehrlich’s techniques in staining blood films and differential blood cell counting in 1879, the narrative highlights key milestones in the establishment of hematology as a distinct field of medicine. The contributions of various scientists, including Antoni van Leeuwenhoek, Alfred Donne, and Gabriel Andal, collectively shaped our current knowledge of blood cells.
Conclusion
The blood cells provides a rich tapestry of information encompassing their structure, functions, and associated disorders. It serves as a comprehensive resource for understanding the dynamic nature of blood and the pivotal roles played by red blood cells, white blood cells, and platelets in maintaining homeostasis within the human body. The integration of historical discoveries adds depth to the narrative, highlighting the continuous quest for knowledge that has defined the field of hematology. This article not only serves as an educational tool but also showcases the remarkable advancements in medical science and technology that have propelled our understanding of blood cells to new heights. As we unravel the mysteries of hematopoiesis, we gain valuable insights into the machinery that sustains life within our veins.
Medical students encounter significant academic challenges during their studies, balancing coursework, clinical rotations, research, and personal commitments. Expert Academic Assignment Help offers tailored assistance to meet their needs, providing study materials, tutoring, assignment help, and exam preparation. Beyond academics, it fosters a supportive environment for mentorship and guidance. In essence, Expert Academic Assignment Help is a valuable resource for medical students, empowering them to excel academically and develop into competent healthcare professionals. Contact us at [email protected] for professional assistance.
#assignment help#medical students#healthcare#nursing school#nursing student#medicine#medical help#academic assignments#medical university#university student#university#university life#medical student#student life#student#nursing#nurse#pharmacy colleges#college student
1 note
·
View note
Text
Taking Charge of Your Health: Booking a CBC (Complete Blood Count) Online

Ever felt that nagging feeling that something might be off with your health? Fatigue, unexplained weight loss, or persistent infections could be signs your body is trying to tell you something. A complete blood count (CBC) is a simple yet powerful tool that can offer valuable insights into your overall health.
This blog post will guide you through everything you need to know about CBC tests, including:
What is a CBC test and what does it measure?
Why might you need a CBC test?
Benefits of booking a CBC online
How to book a CBC online (step-by-step guide)
Understanding your CBC results
What is a CBC Test and What Does it Measure?
A CBC is a routine blood test that assesses the overall health and function of your blood cells. It provides a detailed picture of three main types of blood cells:
Red blood cells (RBCs): These cells carry oxygen throughout your body. The CBC measures the number of RBCs, their size and shape, and the amount of hemoglobin (the protein in RBCs that carries oxygen).
White blood cells (WBCs): These cells are the body's defense system, fighting infections and illnesses. The CBC measures the total WBC count and the number of different types of WBCs, such as lymphocytes, neutrophils, monocytes, eosinophils, and basophils.
Platelets: These are tiny cell fragments that help blood clot. The CBC measures the platelet count, which is important for proper blood clotting and preventing excessive bleeding.
Why Might You Need a CBC Test?
Doctors often order a CBC test for various reasons, including:
Routine health checkups: A CBC is a standard part of many preventive care checkups, helping to monitor your overall health and identify potential issues early on.
Unexplained symptoms: Symptoms like fatigue, fever, weakness, or weight loss can prompt a doctor to order a CBC to investigate the underlying cause.
Suspected medical conditions: A CBC can help diagnose various conditions like anemia (low red blood cell count), infections (elevated white blood cell count), or bleeding disorders (low platelet count).
Monitoring treatment effectiveness: If you're undergoing treatment for a condition that affects your blood cells, such as chemotherapy or radiation therapy, a CBC can help monitor the effectiveness of the treatment.
Benefits of Booking a CBC Online
In today's fast-paced world, convenience is key. Booking a CBC online offers several advantages:
Save Time: Skip the waiting room! Booking online allows you to schedule your test at a convenient time without long wait times at the lab.
Accessibility: Many online lab services offer home sample collection, eliminating the need to travel to a lab facility.
Cost-Effectiveness: Online lab services often provide competitive pricing compared to traditional lab visits.
Transparency: Many online platforms clearly display test costs and turnaround times upfront, allowing you to plan accordingly.
How to Book a CBC Online (Step-by-Step Guide)
Booking a CBC online is a simple and convenient process. Here's a step-by-step guide:
Find a Reputable Lab Service: Conduct a quick online search for "book CBC test online" or "online lab testing CBC." Look for established labs with positive customer reviews and accreditations from recognized organizations.
Review Services Offered: Ensure the lab offers CBC testing as part of their services. Some labs may provide additional blood tests at discounted packages.
Schedule Your Test: Once you've chosen a lab, navigate to their website and find the booking section. Select "CBC" or "Complete Blood Count" from the list of available tests.
Choose Your Location: Most online labs offer in-lab testing or home sample collection. Select the option that best suits your needs.
Provide Personal Information: Fill out the online form with your name, contact details, insurance information (if applicable), and a doctor's referral (if required).
Payment and Confirmation: Review the test cost and choose your preferred payment method. Once payment is complete, you'll receive a confirmation email with additional instructions.
Understanding Your CBC Results After your blood test, the lab will analyze your sample and send you the results electronically or through a secure online portal. Your results will detail the values for each component measured in the complete blood count (CBC), along with a reference range indicating the normal levels for healthy adults. It's important to remember that interpreting your CBC results should be done by a qualified healthcare professional. While some variations from the reference range might be normal for you. Book complete blood count (CBC) online
0 notes
Text
Complementary therapies for autism/Autism Spectrum Disorder (ASD) - Overview and recommendations for biological therapies

Autism spectrum disorder (ASD) is a genetically based neurodevelopmental disorder characterized by persistent impairments in social interaction and restricted, repetitive patterns of behavior, interests, and activities.
它是一种因大脑区域功能障碍而引起的精神障碍。
这也是一种精神表现异常的综合症。
It is a neurodevelopmental disorder of neural connections.
Excluding brain trauma, infection, tumors, etc., most causes of ASD are caused by inherited metabolic disorders (IEMs).
1. Signs and symptoms of ASD
Listed below are some common types of behaviors seen in people with autism/ASD . Not all people with autism will exhibit all of the behaviors, but most will exhibit some of the following.
1. Social communication/interaction behaviors may include:
Little or infrequent eye contact
Does not seem to be looking at or listening to others
Rarely shares interest, affection, or enjoyment of objects or activities (including rarely pointing to or showing things to others)
No response or slow response to others' name or other verbal requests for attention
Difficulty in back-and-forth conversation
Frequently talks at length about favorite topics without noticing that others are not interested or without giving others a chance to respond
Displaying facial expressions, movements, and gestures that are inconsistent with what is being said
Unusual intonation, which may sound sing-song or flat and robotic
Difficulty understanding other people’s perspectives, or being unable to predict or understand other people’s behavior
Difficulty adjusting behavior to social situations
Difficulty engaging in imaginative play or making friends
2. Restrictive/repetitive behaviors may include:
Repeating certain behaviors or having unusual behaviors, such as repeating words or phrases (a behavior called echolalia)
A persistent interest in a particular subject, such as numbers, details, or facts
Shows an exaggerated interest, such as in moving objects or parts of objects
Feeling uneasy about minor changes in daily life and having difficulty adjusting to transitions
Being more or less sensitive than other people to sensory input (such as light, sound, clothing, or temperature)
3. Complications
ASD is often associated with multiple physical and psychiatric disorders:
Feeding problems: eating disorders, anorexia nervosa, aversion to certain types of food, only eating foods with a certain taste/texture, which may cause abnormal weight, gastrointestinal symptoms such as diarrhea and constipation, or lack of nutrition due to unreasonable eating
Sleep disorders: sleep anxiety, tossing and turning, difficulty falling asleep, inability to lie flat to sleep, insomnia, repeated awakenings at night, sleepwalking and other abnormal sleep structures
Gastrointestinal (GI) problems: Gastroesophageal reflux, eosinophilic esophagitis, nausea and vomiting, constipation, diarrhea, flatulence, colitis, food intolerances
Allergic diseases: asthma, nasal allergies, allergic diseases (IgE-mediated), food allergies and intolerances
Toileting problems: difficulty learning to use the toilet during the day and night, not knowing when to use the toilet, communicating the need to use the toilet, being able to use the toilet independently or promptly, learning to use different and unfamiliar toilets, self-wiping, sensory differences (dislike of noises made by the toilet, the sensation of urine/faeces, cold toilet seats or excessive concern about water in the toilet), fecal smearing, a range of specific continence difficulties including bowel or bladder control difficulties
Abnormal responses to sensory stimuli: Over-, under-, or abnormal responses to environmental stimuli such as noise, touch, smells, tastes, or visual stimuli. Examples include looking out of the corner of the eye, excessive focus on edges, spinning objects, shiny surfaces, lights, or smells, refusing or only eating foods with certain tastes/textures, obsessive sniffing or licking of non-food objects, tactile defensiveness or resistance to being touched, or hypersensitivity to certain types of touch; light touch may be painful, while deep pressure may be soothing. There may be resistance to wearing certain textures or colors of clothing against the skin, apparent indifference to pain, a strong preference and/or persistence for certain textures and a strong aversion to other textures, and hypersensitivity to certain frequencies or types of sounds (e.g., a fire truck in the distance) but no response to nearby sounds or sounds that would scare other children.
For more information about this type of auditory and visual abnormalities, please refer to previous articles:
Symptoms of autism and CVI cranial neurological visual impairment
Mental health problems: epilepsy, intellectual disability, learning disabilities, attention deficit hyperactivity disorder (ADHD), tics, anxiety and depression, obsessive-compulsive disorder (OCD), personality and mood changes, visual and auditory hallucinations, phobias, schizophrenia and bipolar disorder.
4. Other clinical features:
Motor deficits: Children with ASD may have motor deficits, including abnormal gait, clumsiness, walking on tiptoes, or other signs of abnormal movement, such as low muscle tone. Motor deficits are common but are not a key feature of ASD.
Head deformity : About 15% of children with ASD have microcephaly, which is common in people with related diseases (such as Angelman syndrome and Smith-Lemli-Opitz syndrome). Among children with ASD, about 1/4 have head circumference greater than the 97th percentile. Up to 70% of children with ASD have accelerated head growth in the first year after birth, but these children do not necessarily become macrocephalic. People with ASD and macrocephaly may have PTEN gene mutations and are therefore at risk of developing hamartoma syndrome.
Special skills : Some people with ASD have special skills (i.e., "scholar" skills) in areas such as memory, mathematics, music, art, or puzzles, even though they have significant deficits in other areas. Other special skills include calendar calculation (calculating the day of the week on a given date) and precocious reading (spontaneous and premature mastery of single-word reading). This reading is usually formal, with little comprehension of meaning or understanding of the purpose of reading.
2. Causes of ASD: Impaired brain function
Excluding brain damage caused by brain trauma, tumors, and infectious diseases, most brain functional abnormalities in ASD patients are caused by IEM, an inherited metabolic disorder.
The introduction to Brodmann's brain partitioning shows the functions of various brain areas. When a patient has a problem in a certain area, the function of that area will be abnormal. Some areas are connected throughout the functional areas. Damage to these areas will cause the communication and expression of information in several areas.
The functional areas of the brain include the intelligence area, mental thinking and emotional area, muscle control, limb movement area, etc. These functional areas are interconnected with the eyes, ears, and body perception, and transmit the information seen by the eyes, heard by the ears, and felt by the body (cool, painful, hot and other limb sensory signals) to the various functional areas of the brain for analysis and response, and then respond to commands. This is accomplished by a whole set of brain functions. The neurons in some areas are interconnected and none of them can be missing.
This type of disease affects different areas of the brain in some patients, so the symptoms shown by each patient are different:
Some patients have normal or superior intelligence and logical thinking, but are not good at handling interpersonal relationships, that is, they lack emotional intelligence.
Some patients have normal intelligence but impaired logical thinking, and will show excellent stereotyped memory, but poor logical ability such as mathematical thinking.
Some patients have normal brain functions elsewhere, only poor sense of direction and prosopagnosia, which do not affect their daily life and work. However, they may have consumed large amounts of food and alcohol that their bodies cannot metabolize during adolescence or an infection, which triggered a metabolic crisis and caused brain damage. Therefore, if a metabolic disease exists, even if it is only a slight influence, it is still necessary to diagnose the disease and conduct disease management.
Articles that affect brain function:
Brodmann area
Brodmann area
Visual information travels through two communication streams: the dorsal stream and the ventral stream
3. Causes of ASD: Inherited Metabolic Disorders (IEM)
1. Common types of inherited metabolic disorders (IEM) :
Small molecule diseases: (1) Protein (amino acid) metabolism disorders and organic acidemias ; (2) Carbohydrate metabolism disorders ; (3) Fatty acid metabolism disorders ; (4) Porphyrin and heme metabolism disorders ; (5) Mineral absorption and transport disorders .
Organelle diseases : (1) Lysosome and lysosome-related organelle diseases; (2) Peroxisome diseases ; (3) Mitochondrial encephalomyopathy .
Others: (1) Purine, pyrimidine and neurotransmitter metabolism disorders ; (2) Vitamin and non-protein cofactor metabolism and transport disorders ; (3) Energy metabolism disorders ; (4) Cholesterol synthesis and metabolism disorders ; (5) Creatine metabolism disorders
For the types and diagnosis of IEM, please refer to previous articles:
Types and diagnostic methods of inherited metabolic disorders (IEMs)
Pathogenesis and clinical features of inherited metabolic disorders (IEMs)
Metabolic emergencies in SWAN syndrome (suspected inherited metabolic disorder): Presentation, evaluation, and treatment
Blood ammonia, blood sugar, blood lipids, lactic acid, blood gas analysis, cerebrospinal fluid analysis
2. Biochemical signs and symptoms of IEM
The signs and symptoms are grouped into organ systems and conditions (autonomic, cardiovascular, dental, dermatologic, digestive, malformation, ear, endocrine, eye, genitourinary, hair, blood, immune, metabolic, muscular, neurological, psychiatric, renal, respiratory , skeletal, neoplastic, etc.)
2.1 Eating disorders nervosa (anorexia and dietary avoidance):
Stereotyped and monotonous eating habits ;
Avoidant/Restrictive Food Intake Disorder (ARFID) ;
Mitochondrial neurogastrointestinal encephalomyopathy ( MNGE)
Migraine Complications: Irritable Bowel Syndrome (IBS)
2.2 Epileptic disorders: Epileptic disorders caused by IEM
2.3 Phenotypes of cerebral palsy: spastic type (including diplegia, hemiplegia and quadriplegia), involuntary movement type (including dystonia and choreoathetosis), ataxia type
2.4 Mental disorders: depression, autism, cognitive impairment, Alzheimer's disease
2.5 Movement disorders: dystonia, myoclonus, chorea, tremor, tics, Parkinsonism or ataxia, eye movement disorders, progressive myoclonic epilepsy, stroke and stroke-like seizures
2.6 Metabolic myopathy: muscle weakness, hypotonia, exercise intolerance, myoglobinuria (soy sauce urine), rhabdomyolysis
2.7 Metabolic kidney diseases: renal tubules, glomeruli, renal cysts, kidney stones, renal malformations
2.8 Metabolic liver disease MLD: Hepatomegaly; hepatocellular disease with elevated transaminases or frank acute liver failure; cholestasis; steatosis; fibrosis or cirrhosis; and liver tumors
2.9 Gastrointestinal disorders: loss of appetite, gastroesophageal sphincter dysfunction, constipation, dysphagia, vomiting, gastroparesis, intestinal pseudo-obstruction, diarrhea, rare manifestations of gastrointestinal cavities, pancreatitis, liver disease
2.10 Respiratory system diseases: interstitial lung disease, lower respiratory tract infection, chronic airway aspiration, pulmonary hypertension, alveolar hypoventilation, upper airway obstruction
2.11 Immunodeficiency: Innate and Adaptive Immunity
2.12 Metabolic ear disease: sensorineural hearing loss, mixed hearing loss, congenital external ear anomaly, congenital external ear anomaly, inner ear or retrocochlear involvement
2.13 Metabolic eye diseases: retinitis pigmentosa (RP), choroideremia, Stargardt's disease, cone-rod dystrophy (CRD), Leber congenital amaurosis (LCA)
2.14 CVI cortical visual impairment (cortical blindness): agnosia, visual field loss, visual stimulation
2.15 Tumors: Accumulation of toxic metabolites, tumor metabolites, mitochondrial dysfunction, metabolic reprogramming
2.16 Metabolic skin diseases and hair abnormalities: papules, skin nodules, xanthomas, melanin pigmentation, photosensitive dermatitis, pellagra, acrodermatitis enteropathica, angiokeratoma, short, sparse and/or brittle hair, alopecia nodularis
2.17 Metabolic cardiovascular diseases: cardiomyopathy, arrhythmia, vascular disease, cardiometabolic
2.18 Metabolic hypertension: H-type hypertension , endothelial-dependent hypertension
The following collection summarizes this type of genetic metabolic disease series represented by clinical symptoms such as ophthalmology, myopathy, and mental disorders in this collection and continues to update it, hoping to provide some clinical tips for undiagnosed patients. It is hoped that the patients' families will summarize these clues and submit them to professional metabolic doctors for analysis, so as to strive for early diagnosis and standardized treatment, thereby avoiding secondary organ damage caused by the disease, as well as preventing family-related diseases and early warning of reproduction.
Collection:
Inherited metabolic disorders affecting the eyes, liver, kidneys, heart, and muscles
Genetic metabolic diagnostic tests: blood and urine tandem mass spectrometry, blood gas, lactate, blood ammonia, liver and kidney function, etc.
IV. Introduction and evaluation of complementary and alternative therapies (CAM) for ASD
Complementary and alternative medicine (CAM) is defined as: "a diverse group of medical and health care systems, practices, and products that are not generally considered part of conventional medicine"
Traditional treatments for ASD include behavioral therapies, such as intensive behavioral therapies such as applied behavior analysis. Complementary and alternative therapies (biological therapies) are an integrative approach.
Some parents choose CAM (hereafter referred to as biological therapy) because they believe that traditional treatments are ineffective for ASD and they hope to find a cure, while others use CAM as a supplement to traditional treatments to help their children as much as possible. Parents' reasons for choosing CAM may include: treating a variety of symptoms, including the core symptoms of ASD (such as deficits in social communication and social interaction, as well as restricted, repetitive behaviors, interests and activity patterns), inattention, gastrointestinal symptoms and sleep disorders; promoting a healthy lifestyle; and concerns about the safety or side effects of traditional allopathic treatments (such as prescription drugs). Some treatments are used to address the biological conditions that parents believe contribute to ASD, regardless of whether there is scientific evidence to support the hypothesis. Parents who use such treatments have their own circle, which is commonly .
Biological therapies are classified according to their benefits and risks as follows:
1. No benefit
Secretin: Secretin is not recommended for children with ASD. Secretin does not improve the core features of ASD.
Secretin is a gastrointestinal hormone that inhibits intestinal motility and gastric acid release and stimulates the secretion of pancreatic juice and bicarbonate. Secretin may be used to treat children with ASD based on the hypothesis that autism is associated with abnormalities of the gastrointestinal system. However, there is little evidence to support this hypothesis. A 2012 systematic review of 16 randomized controlled trials involving more than 900 children failed to confirm that secretin improves the core features of autism. No serious side effects have been reported.
Facilitated communication: Facilitated communication is not recommended for communicating with children with ASD.
In assistive communication technology, a helper physically guides a nonverbal child to communicate using an output device (such as a keyboard or mouse) with their hands. An analysis of published research on assistive communication found no evidence to support its use for people with communication disabilities. Controlled studies show that it is the helper, not the child, who provides the information.
Augmentative communication must be distinguished from augmentative communication, which is an established nonverbal method of communication that uses gestures, picture exchanges, transducers, or sound output devices to communicate without the help of a facilitator. Augmentative communication is appropriate for some children with ASD.
2. Potentially beneficial but with potential risks
Gluten-free and casein-free diet: A gluten-free and casein-free (GFCF) diet is not recommended for children with ASD unless celiac disease or gluten sensitivity is confirmed. Evidence for the efficacy of a GFCF diet is weak and limited; strict adherence to a GFCF diet is difficult and may result in nutritional deficiencies (eg, calcium, vitamin D, amino acid deficiencies) unless monitored by a registered dietitian.
Despite the lack of evidence, many caregivers put children with ASD on a GFCF diet. Children with ASD need to get enough vitamin D, calcium, and protein. Rice milk, almond milk, and potato milk do not provide adequate protein. (See: Nutrient Protein )
The rationale for the use of a GFCF diet in children with ASD is that increased intestinal permeability allows gluten and casein peptides to leak from the intestine, resulting in excessive opioid activity and thus contributing to the behaviors seen in ASD. However, most children with ASD do not have an increased incidence of celiac disease or excess opioid compounds in their urine.
Multiple systematic reviews evaluating GFCF diets in children with ASD using randomized trials have found limited and weak evidence supporting the benefits of GFCF diets. Limitations include lack of blinding, small sample sizes, and differences in patient populations, interventions, and outcome measures.
A meta-analysis of six randomized trials with a total of 143 participants found that children following a GFCF diet had similar physician-reported core ASD symptoms, caregiver-reported levels of functioning, and behavioral disturbances compared with children following a conventional diet. The quality of this evidence was limited due to inconsistency, imprecision, and risk of bias. A subsequent randomized trial of 66 children aged 36 to 69 months found no differences in autism symptoms, maladaptive behaviors, and intelligence after following a study diet for 6 weeks.
Therefore, the GFCF diet may be effective for ASD patients with celiac disease and gluten allergy. Therefore, it is recommended that such patients can use a gluten-free diet. Of course, before using a gluten-free diet, it is necessary to test whether there is celiac disease or gluten allergy.
Symptoms of Celiac Disease Diagnosis and Gluten-Free Diet Introduction:
Diagnosis of Celiac Disease
Gluten-free diet
3. Unclear benefits and possible risks
The treatments described in this section have little evidence of effectiveness and may be harmful. Using these treatments may divert time, energy, and money from proven treatments. Given their potential harm, their use in treating ASD is discouraged.
Intravenous immune globulin: Intravenous immune globulin (IVIG) is not recommended for the treatment of ASD unless there is another indication for IVIG, such as a proven immunodeficiency or if the patient has immunodeficiency caused by an inherited metabolic disorder (IEM).
Immune deficiency caused by inherited metabolic diseases: innate immunity and adaptive immunity
The use of IVIG and other immunotherapies in children with ASD is based on the hypothesis that ASD is associated with a dysregulated immune system. Data to support this hypothesis are limited. Although small, open-label trials have shown that IVIG improves some areas in children with ASD, the available evidence is mixed, and adverse effects are common. The only randomized, placebo-controlled, blinded trial of 12 males with ASD found similar physician-rated outcomes in the IVIG and placebo groups, but greater improvements from baseline in scores on the Aberrant Behavior Checklist (ABC) or some subscales of the symptom inventory were observed in the IVIG group; the researchers noted that IVIG may only be beneficial in certain patients and cautioned against its misuse.
IVIG is expensive. Potential adverse effects of IVIG and other immunotherapies include transmission of blood-borne pathogens.
Chelation therapy: Chelation therapy is not recommended for ASD patients without a confirmed metal metabolism disorder. Chelation therapy has no evidence of efficacy and may cause serious harm.
Chelation therapy is used in metal metabolism disorders. Patients with such disorders will show abnormal manifestations of autism. Therefore, chelation therapy has flowed into the biosphere. Some parents do not diagnose ASD children with metal metabolism disorders and use chelation therapy, which is highly unrecommended. Moreover, the diagnosis of metal metabolism disorders is rigorous. It is necessary to test the excretion and metabolism of metal trace elements in patients (hair, blood, urine) in regular and specialized hospitals, and also to conduct genetic diagnosis, rather than diagnosing by measuring trace elements in fingertip blood in some institutions.
Metal metabolism disorders: Copper metabolism disorders: Wilson's disease, Menkes' disease, Iron metabolism disorders: familial hyperferremia, neurodegeneration with brain iron deposition, Magnesium metabolism disorders: magnesium-dependent epilepsy, magnesium transporter mutation disease, magnesium malabsorption syndrome, Manganese metabolism disorders: hypermanganeseemia.
Introduction to the diagnosis of metal metabolism disorders: Metal metabolism disorders and chelation therapy
Chelation is the process of removing heavy metals from the body by administering a substance such as ethylenediaminetetraacetic acid (EDTA), 2,3-dimercaptosuccinic acid (DMSA), or 2,3-dimercapto-1-propanesulfonate (DMPS).
The rationale for using chelation to treat ASD is based on the hypothesis that the behaviors of children with ASD are secondary to mercury or other heavy metal poisoning that these children are unable to effectively excrete. However, there is little evidence to support this hypothesis. The clinical manifestations of mercury poisoning do not resemble those of ASD, and there is no evidence to support a causal relationship between thimerosal and autism.
The safety and efficacy of chelation therapy (including over-the-counter oral or rectal products) for ASD have not been adequately studied in controlled trials. The only randomized trials have methodological limitations. Chelating agents bind ions nonspecifically and, in addition to reducing toxic heavy metals, can also reduce calcium, iron, and magnesium levels. Close monitoring is required when chelating agents are used to treat proven heavy metal poisoning. Fatal hypocalcemia has been reported with chelation therapy using sodium EDTA (but not calcium EDTA).
Hyperbaric oxygen: Hyperbaric oxygen therapy (HBOT) is not recommended for the treatment of ASD. HBOT is expensive and the evidence of its effectiveness is not sufficient.
The rationale for using HBOT to treat autism is that increasing atmospheric pressure increases the amount of oxygen delivered to the brain, thereby reducing swelling and promoting brain recovery.
A 2016 systematic review included the only randomized trial with 60 children and found that HBOT did not improve social interaction, behavioral problems, communication, and cognitive function in children with ASD. A subsequent review included literature from 2015-2021 and concluded that the effect of HBOT on children with ASD has not been proven. Adverse effects of HBOT include: barotrauma of the middle ear, sinuses, or lungs, reversible myopia, pulmonary oxygen toxicity, and seizures.
Antimicrobial agents: We do not recommend the use of antimicrobial agents, including antifungals (eg, nystatin, fluconazole), antibiotics, or antivirals, for the treatment of ASD. These agents have unproven efficacy and may cause adverse effects.
The use of antimicrobial agents is based on the hypothesis that individuals with ASD have an imbalance in the gut microbiota, such as overgrowth of yeast or bacteria.
Few controlled trials have evaluated antimicrobial therapy in children with ASD. Adverse effects include hypersensitivity reactions, hepatotoxicity, anemia, diarrhea, exfoliative dermatitis, and promotion of antimicrobial resistance.
Vitamin B6 and Magnesium: Vitamin B6 (pyridoxine) and magnesium supplements are not recommended for the treatment of ASD.
Although vitamin B6 and magnesium (magnesium is added to reduce the side effects of vitamin B6) have long been used to treat psychiatric disorders, few high-quality studies have specifically evaluated vitamin B6-magnesium supplementation for ASD. A 2010 systematic review included three small randomized trials (n = 33) with methodological flaws and found that the current evidence was inconclusive.
High doses of vitamin B6 (>100 mg/d) may cause neuropathy.
Vitamin B6 and magnesium are clinically used to treat some inherited metabolic disorders IEM. For example, magnesium is used to treat magnesium-dependent epilepsy and migraine, and vitamin B6 is used to treat vitamin B6-dependent epilepsy and hydroxykynureninuria, among which patients with hydroxykynureninuria have autistic symptoms.
Familial hypomagnesemic epilepsy is a hereditary disease of epilepsy and hypomagnesemia caused by mutations in magnesium transporters. The cause is due to mutations in magnesium transporters (MgT1) or magnesium transporters 2 (MgT2), which are responsible for the absorption and excretion of magnesium in the intestines and kidneys. Clinical manifestations include epilepsy, hypomagnesemia, hypocalcemia, and increased .
Neurotrophic drugs: mouse nerve growth factor, brain protein hydrolysate injection, etc. are promoted as neuroprotectants for treating brain nerves, but they cannot improve the symptoms of ASD because these neuroprotectants cannot penetrate the blood-brain barrier, and most ASDs are caused by genetic metabolic disorders. Repairing brain nerves directly without diagnosing the specific cause and resolving these metabolic disorders is like the broken barrel effect of patching while leaking.
Fecal microbiota transplantation (FMT): There is a hypothesis that the gut-brain axis causes autistic symptoms. Such diseases can be treated by improving the intestinal flora of autistic patients. However, some autistic patients do not see improvement in their symptoms after such treatment. There is currently insufficient evidence to prove this hypothesis. For example, some studies have shown that intestinal flora disorders are caused by the stereotyped diet of ASD patients. Some IEM diseases can cause the production of intestinal ammonia and propionic acid, which in turn cause a series of gastrointestinal symptoms such as abdominal pain, vomiting and refusal to eat. Therefore, some ASD patients have such intestinal flora imbalance and eating disorders caused by IEM diseases. Therefore, the disease should be diagnosed and treated instead of treating such abnormal gastrointestinal symptoms. A radical treatment method is needed.
4. Other interventions
Other interventions that have not been shown to be beneficial for ASD and their potential adverse effects include:
Vitamin A: Taking large amounts of vitamin A can cause toxicity
Vitamin D: A small randomized trial showed that vitamin D supplementation did not improve the core symptoms of ASD. Excessive use of vitamin D can also have adverse effects
Homeopathy: May contain undeclared toxic ingredients
Vagus nerve stimulation: It is a recognized intervention for epilepsy, but its effectiveness in ASD has not been proven.
Stem cell transplantation is currently being studied to treat certain central nervous system diseases that require nerve regeneration, such as spinal cord injury. However, since most ASDs are caused by genetic metabolic disorders, it is physiologically meaningless to use stem cells directly for regenerative treatment without confirming the specific cause. In addition, stem cell transplantation to repair nerve damage is still in the research stage and has not yet entered clinical treatment.
picture
Given the lack of demonstrated benefit and potential for harm, these therapies are not recommended for individuals with ASD.
5. Possibly beneficial and low risk
With the exception of melatonin for sleep disturbances, these therapies are not strongly encouraged for ASD given the lack of clear benefit. If used, these therapies should be monitored for side effects, interactions, and impact on the prescribed/recommended treatment.
Music therapy: Music therapy may be beneficial for children with ASD and is unlikely to be harmful. Until further research is done, we do not strongly encourage the use of music therapy to treat core features of ASD unless it is included in a broader comprehensive behavioral treatment program.
The use of music therapy for children with ASD is based on the hypothesis that engaging in musical interaction (sometimes interpreted as a form of nonverbal or preverbal language) can help individuals with autism develop communication skills and social interaction abilities.
A systematic review of 26 clinical trials with 1165 participants comparing music therapy with standard care or 'placebo' treatment (similar treatment that does not include music) found that music therapy was likely to improve participants' overall condition soon after the end of the intervention, with improvements in overall autism severity and quality of life, and did not appear to increase adverse events, with moderate confidence. It is uncertain whether music therapy improves social interaction, non-verbal communication, and verbal communication. Although no differences were found between the groups, the quality of the evidence was low due to risk of bias and lack of precision.
Melatonin: Open observational studies and randomized placebo-controlled trials have found that melatonin is beneficial for falling asleep and maintaining sleep in children with ASD. A meta-analysis included 5 randomized crossover trials with a total of 57 children with ASD. Compared with placebo, melatonin prolonged sleep time by 73 minutes and shortened sleep latency by 66 minutes, but did not affect nighttime awakenings. It has very few side effects. In a single study, the melatonin dose was 0.75-10 mg/d, and the duration of treatment was 14 days to 6 months.
These results suggest that melatonin 1-10 mg given 30 minutes before bedtime to children with ASD may help with sleep onset and sleep duration in the short term (i.e., up to 6 months). However, there are no guidelines for its use, nor is there information on long-term use and side effects. Side effects of melatonin may include difficulty awakening, daytime sleepiness, and enuresis.
If children with ASD have difficulty falling asleep and staying asleep despite appropriate sleep hygiene and behavioral or environmental interventions, melatonin is recommended. The starting dose of melatonin is usually 0.5 to 1 mg (depending on age), which can be increased by 1 mg each time as needed, to a maximum dose of 10 mg (although higher doses may be used clinically).
Melatonin is an over-the-counter drug that is not regulated by the FDA. When parents or caregivers purchase melatonin, they should choose preparations that contain melatonin as the only active ingredient.
Oxytocin: Small or open studies have shown that oxytocin may improve social interaction or function in children with ASD, but a blinded randomized trial did not find any benefit. Further randomized trials are needed to determine whether oxytocin can be recommended for improving social interaction in children with ASD.
Therapeutic Horseback Riding: Therapeutic horseback riding (hippotherapy) is not actively encouraged as a core treatment for children with ASD, but may be useful if used within a broader comprehensive behavioral/educational treatment program and monitored appropriately. Although some evidence suggests that hippotherapy is beneficial, further research is needed before this therapy can be actively recommended.
It has been hypothesized that therapeutic horseback riding may stimulate multiple domains of functioning in children with ASD, such as cognition, socialization, and gross motor skills. A randomized trial of 116 children and adolescents with ASD compared 10 weeks of therapeutic horseback riding with 10 weeks of barn-based activities without horseback riding and showed that therapeutic horseback riding improved measures of hyperactivity and irritability after approximately 5 weeks. Parent-report assessments showed that therapeutic horseback riding also improved social cognition and social communication. In a smaller nonrandomized study, 19 children with autism who participated in 12 weeks of therapeutic horseback riding had improvements in attention, distractibility, and social motivation compared with 15 wait-list controls.
The risk of injury from therapeutic horseback riding is similar to that of other "limited contact" recreational activities (eg, baseball, ice skating). If caregivers choose this type of therapy, they must emphasize the use of helmets and appropriate supervision.
Other types of animal/pet therapy: Observational studies suggest that the presence of animals can reduce social stress and increase social behavior in individuals with ASD, but further research is needed before animal therapy can be recommended.
A preliminary randomized trial comparing canine-assisted occupational therapy with standard occupational therapy (wait-list control) in 22 children with ASD found a trend toward improved on-task behavior and goal achievement in the canine-assisted occupational therapy group, but more research is needed.
Sulforaphane: Sulforaphane supplementation is not recommended for children with ASD. Further research is needed to confirm the benefits of sulforaphane.
Sulforaphane is an antioxidant derived from broccoli sprout extract that increases the activity of specific genes to protect aerobic cells from oxidative stress, inflammation, and DNA damage. It has been hypothesized that sulforaphane may reverse abnormalities associated with ASD, including neuroinflammation, oxidative stress, and decreased glutathione synthesis, mitochondrial function, and oxidative phosphorylation.
One trial evaluated the effect of sulforaphane in ASD, in which young men aged 13 to 27 years were randomized to receive daily sulforaphane (n=29) or placebo (n=15) for 18 weeks. During the treatment period, the ABC, Social Responsiveness Scale, and Clinical Effectiveness Summary-Improvement Scale rated by parents and doctors showed that the behavior of the sulforaphane group improved compared with baseline. After discontinuation of sulforaphane, the subject's behavior returned to baseline. A subsequent placebo-controlled randomized trial included 57 children with ASD aged 3 to 12 years, with 45 followed after 15 weeks (22 in the sulforaphane group and 23 in the placebo group). Clinical effects varied by outcome measure (no benefit on the Ohio Autism Clinical Global Rating Scale and Social Responsiveness Scale 2nd Edition, improvement on the ABC Scale) and were less significant than in the trial in young men. Sulforaphane was well tolerated in both trials, but the sample sizes were too small to adequately assess adverse effects. While these results are encouraging, more research is needed.
Until further research is done, broccoli sprouts or other foods rich in sulforaphane (such as Brussels sprouts and cauliflower) would not be specifically recommended for children with ASD, but given their other health benefits, there is no objection to their consumption. The subjects consumed 50-150 μmol of sulforaphane, which is much higher than the amount that can be obtained from food.
Transcranial magnetic stimulation: Transcranial magnetic stimulation (TMS) is not recommended for children with ASD unless used as a clinical trial protocol. Although TMS appears to be safe in clinical trials, further research is needed before TMS can be recommended for the treatment of children with ASD.
TMS is an energy-based therapy that may work through electromagnetic induction, which can alter neural excitability. Controlled and uncontrolled studies have shown that TMS may improve depression, bipolar disorder, schizophrenia, epilepsy, and Tourette syndrome.
The use of TMS in children with ASD is based on the hypothesis that autism is associated with disturbances in cortical modularity. A 2016 literature review found some evidence that TMS may reduce symptoms of ASD (core and associated symptoms), but more rigorous research is needed. TMS is generally considered safe for use in children and adults. Seizures are the most serious adverse event of TMS, but they are very rare.
Other interventions:
Other interventions that lack clear scientific evidence to support benefits for children with ASD but are unlikely to be harmful include:
●Yoga.
●Body manipulation and energy therapies such as Chi massage, therapeutic touch, healing touch, Reiki.
●Biofeedback/neurofeedback – In biofeedback/neurofeedback, behavioral therapy focuses on achieving self-regulation of cortical electrical activity measured by electroencephalography (EEG), often with the use of visual feedback. Reported side effects are minimal but may include headaches. Although safe, neurofeedback can be expensive and has not been proven to improve core symptoms of ASD.
●Hypnosis therapy.
Vitamin C (up to 2 g/day).
●Vitamin B12.
●Folinic acid and folic acid.
Given the lack of clear evidence of benefit, we discourage the use of these interventions in individuals with ASD unless the individual has a documented inherited metabolic disorder.
6. Benefits are unclear but risks are low
Given the unclear benefits of these therapies, their use in the treatment of ASD is discouraged. If caregivers insist on using these therapies, it is recommended that they monitor for side effects, interactions, and effects on the prescribed/recommended treatment.
Auditory Integration Training: Auditory Integration Training (AIT) is discouraged for children with ASD. Although AIT has a low risk of adverse effects, its efficacy is unproven and it is expensive.
The use of AIT in children with ASD is based on the hypothesis that repeated exposure to changing sounds through headphones can functionally alter central auditory processing, affecting language and behavior.
A systematic review of randomized trials showed that AIT did not improve language outcomes in children with ASD. The evidence is limited by inconsistent results and small sample sizes.
Omega-3 fatty acids: Although they may have some benefits, omega-3 fatty acids are not recommended for treating the core symptoms of ASD. Omega-3 fatty acids are essential fatty acids that may have cardiovascular benefits and include eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA). Some studies have shown reduced plasma omega-3 fatty acid concentrations in children with ASD, but no clear clinical association has been found.
Introduction to fats: Energy-supplying nutrients for the human body: fatty acids and MCT oil
Several systematic reviews have evaluated the use of omega-3 fatty acids for ASD through randomized trials, with inconsistent results. Most trials found that omega-3 fatty acid supplementation did not improve core symptoms or associated symptoms of ASD, but was unlikely to be harmful. A meta-analysis showed that omega-3 fatty acid supplementation could slightly improve language and social deficits and related symptoms (such as inattention, irritability, behavioral disorders, and cognitive impairment), but these results were limited by the low quality of the included studies.
Common side effects of omega-3 fatty acid supplementation include gastrointestinal upset, such as nausea and diarrhea. Studies evaluating omega-3 fatty acid supplementation in individuals with ASD have not reported major side effects. Omega-3 fatty acid supplementation should be used with caution in patients with bleeding disorders or those with fish allergies.
There are no specific dosing guidelines for omega-3 fatty acids. Studies in children with ASD have used doses of 1.3 g/d and 1.5 g/d.
Probiotics: It is hypothesized that probiotics can correct the imbalance of intestinal flora in individuals with ASD. FDA regulation of probiotics depends on the type of product, such as biologics, drugs, dietary supplements, medical foods, and food ingredients. Probiotic products that are not strictly regulated may contain undeclared ingredients or strains.
Although the relationship between intestinal flora and ASD remains a hot topic and related research is underway, there is insufficient evidence to determine whether there is a causal relationship between the two and which is the cause and which is the effect. For example, studies have suggested that restricted diet and eating disorders in ASD patients affect the intestinal flora of many ASD people, rather than the intestinal flora causing ASD symptoms.
Eating disorders nervosa (anorexia and food avoidance):
Stereotyped and monotonous eating habits ;
Avoidant/Restrictive Food Intake Disorder (ARFID) ;
Mitochondrial neurogastrointestinal encephalomyopathy ( MNGE)
Migraine Complications: Irritable Bowel Syndrome (IBS)
The use of probiotics for ASD is discouraged. Although probiotics are unlikely to be harmful, their use in children with ASD has not been rigorously studied. A systematic review of six clinical trials concluded that probiotics have limited efficacy for gastrointestinal or behavioral symptoms in children with ASD. The trials used different strains, concentrations, and durations of treatment. Some evidence suggests that probiotics may be beneficial for other conditions, such as acute gastroenteritis and functional abdominal pain.
Cannabinoids: The use of medical marijuana or cannabinoids for the treatment of ASD or related symptoms is discouraged. Cannabinoids include cannabidiol (CBD; the non-psychoactive component of marijuana), tetrahydrocannabinol (THC; the psychoactive component of marijuana), dronabinol (synthetic THC), etc. Although CBD can be used to treat certain types of childhood epilepsy and appears to be safe, its benefits for children with ASD are not clear.
Although animal models suggest that endocannabinoid signaling may influence genetic disorders associated with ASD (e.g., Fragile X syndrome), few studies have evaluated the direct effects of medical marijuana in individuals with ASD. A randomized trial enrolled 150 individuals aged 5–21 years with ASD and found inconsistent efficacy. The study was limited by the lack of pharmacokinetic data and the wide range of age and functional levels of the participants. Other randomized and open-label trials are currently underway to evaluate the effects of cannabinoids on behavior in children with ASD. Observational studies have found that cannabinoids can lead to subjective improvements in behavioral problems (self-injury, hyperactivity), anxiety, and sleep, and reduce the need for other psychoactive medications. Limitations of these studies include the lack of objective assessment tools, poor follow-up, participant attrition, and inconsistent cannabinoid dosing regimens.
In randomized trials and observational studies of cannabinoid use in individuals with ASD, adverse effects have included somnolence, decreased appetite, irritability, and restlessness. Randomized trials of CBD in children with Lennox-Gastaut syndrome and Dravet syndrome have shown adverse effects including somnolence, fever, decreased appetite, diarrhea, and vomiting. However, some children discontinued CBD because of elevated liver transaminase concentrations.
Other interventions: Other interventions with unclear benefits but unlikely to cause harm include:
●Methylcobalamin and N-acetylcysteine.
●Zinc.
●Herbal products.
●Amino acids, including dimethylglycine (DMG; a derivative of glycine), and other amino acids such as taurine, lysine, and gamma-aminobutyric acid (GABA).
●Digestive enzymes.
Mindfulness-based approaches have been studied for children with ASD and their parents, but the quality of the evidence is low due to methodological limitations (eg, small sample size, use of self-reported outcome measures); mindfulness-based approaches are unlikely to be harmful.
●Acupuncture: Acupuncture is generally safe, but it can cause injury in uncooperative patients, and acupuncture of the brain carries the risk of inducing epilepsy.
●Craniosacral therapy: Although systematic reviews have shown that the risks of craniosacral therapy are low, its adverse effects may be underreported.
●Chiropractic (usually safe for children without spinal abnormalities).
7. Other risks of biological therapies for ASD
Biological therapy is not recommended without analyzing the patient's metabolic disorder range and without the guidance of a doctor and nutritionist, because sometimes the operation is reversed, which can lead to many risks:
Dietary risks: If an autistic patient has ASLD argininosuccinic aciduria (a type of urea cycle disorder), some patients with this disease will show mild intellectual disability and mental abnormalities. Undiagnosed patients will be clinically diagnosed with autism. If this patient follows a gluten-free diet without being diagnosed, the diet will be high in protein. ASLD itself is a protein metabolism problem. This kind of diet will aggravate the patient's metabolic disorder, thereby inducing a metabolic crisis and causing a series of disease risk problems.
Supplement risks: For example, some ASD patients will supplement arginine (an amino acid), but if the patient has hyperargininemia, supplementing arginine is the opposite operation. Some supplement therapies for inherited metabolic disorders (IEM) are used in excess of the instructions. Without a confirmed diagnosis of a specific metabolic disorder, large doses of supplements beyond the instructions, without a doctor's monitoring and management, can cause many health risks. For example, some parents take ASD patients to informal private institutions to test for methyl metabolism disorders, and take large doses of folic acid on the advice of these private institutions. These are not recommended behaviors because the diagnosis of the disease is rigorous. Even if a patient has a certain type of gene mutation during genetic testing, a doctor is needed to check the clinical symptoms with metabolite or enzyme results for diagnosis, and post-diagnosis drug treatment management also requires a doctor's guidance and monitoring.
5. Treatment of IEM diseases and biological treatment of ASD
Most of the alternative/complementary therapies (biological therapies) for ASD are based on IEM therapies. Many of these complementary therapies are not managed by a professional medical system and are basically a kind of therapy that parents in the biological therapy circle communicate privately. Therefore, the treatment is very similar to a hodgepodge therapy. Therefore, it is not recommended that parents carry out this kind of complementary therapy without the guidance of a professional nutritionist or doctor. ASD patients should not try to explore this kind of complementary therapies like Shennong tasting a hundred herbs, because without the supervision of a doctor, many therapies may bring risks. It is recommended that parents use this kind of therapy and carry out corresponding treatment after the disease is confirmed. For undiagnosed patients, the scope of metabolic disorders of ASD patients can be analyzed under the guidance of professional doctors, as well as corresponding nutritional management.
1. Current treatments for inherited metabolic disorders (IEMs)
1.1 Diet therapy
Low-protein, high-carb diet: For example, maple syrup urine disease restricts leucine, isoleucine, and valine + low-protein, high-carb diet, because maple syrup urine disease is a metabolic disorder of the three amino acids: leucine, isoleucine, and valine. Therefore, by restricting these three amino acids and a low-protein diet, early treatment of patients can achieve normal growth and development.
Dietary management of genetic metabolic disorders: low protein + high carbohydrate + moderate high fat
Ketogenic diet: A diet for GLUT-1 deficiency glucose metabolism disorder and partial mitochondrial metabolism disorder ( PDH ). GLUT-1 deficiency syndrome is a genetic disease (caused by SLC2A1 mutations) characterized by impaired glucose transport across the blood-brain barrier, leading to generalized epilepsy, developmental delay, and related movement disorders. It may also manifest as early-onset absence epilepsy. The ketogenic diet is an effective first-line treatment for this disease, providing ketones to the brain as an alternative source of energy. The ketogenic diet can also provide an alternative energy source for the brain in patients with pyruvate dehydrogenase (PDH) deficiency; PDH deficiency is a mitochondrial disease characterized by lactic acidosis, severe neurological impairment, and occasionally intractable epilepsy. In non-controlled reports, patients who received a ketogenic diet seemed to have improved neurological outcomes.
Dietary therapy for drug-resistant epilepsy and mitochondrial and sugar metabolism disorders: ketogenic diet therapy
Gluten-Free Diet: A gluten-free diet is currently the only way to treat people with celiac disease. Since there are no available medications or therapies, the only way to manage celiac disease is with a strict 100% gluten-free diet. People with non-celiac gluten allergies can also benefit from a gluten-free diet. Celiac disease is an autoimmune disease that is triggered by consuming gluten and causes damage to the small intestine. When someone with celiac disease eats gluten, the immune system sees gluten as a threat and attacks. However, it ends up damaging the villi in the intestines that help digest food. The damaged villi make it nearly impossible for the body to absorb nutrients, leading to malnutrition and many other problems. Mental problems caused by celiac disease: Schizophrenia, autism, brain fog, headaches.
Gluten-free diet
Celiac disease (non-tropical sprue and gluten-sensitive enteropathy)
1.2 Coenzyme factor supplementation
Vitamin B12: Methylcobalamin (a form of B12) is a cofactor for methionine synthase, an essential enzyme that forms methionine from homocysteine. B12 is used to treat methylmalonic acidemia and combined methylmalonic acidemia and homocystinemia.
Coenzymes, enzyme cofactors, and antioxidants that activate metabolic pathways or provide energy for the treatment of inherited metabolic diseases
Mitochondrial cocktail therapy: The term “mitochondrial cocktail” refers to a large number of vitamins, minerals, and other nutrients (L-carnitine) that play an important role in energy metabolism. This type of pharmaceutical supplement is designed to promote key enzymatic reactions, reduce the putative sequelae of excess free radicals, and clear out the toxic acyl-coenzyme A (acyl-CoA) molecules that accumulate in mitochondrial diseases .
Primary mitochondrial disease (PMD) Cocktail therapy of coenzymes, enzyme cofactors, and antioxidants used to activate metabolic pathways or supply energy
Creatine: Creatine is a nitrogenous organic acid that is mainly produced in the kidneys and liver and stored in high-energy demand tissues such as skeletal muscle and brain tissue. Creatine supplementation is often used to treat: creatine deficiency syndrome CDS and some mitochondrial metabolic disorders. CDS is a group of inborn errors of creatine metabolism and transport that cause intellectual disability, behavioral problems, autism, speech delay, epilepsy and movement disorders .
Creatine deficiency syndrome (CDS) causes autism, intellectual disability, intractable epilepsy, and myopathy and movement disorders
Folic acid (vitamin B9): It is necessary for one-carbon transfer reactions; the active form folinic acid (5-formyltetrahydrofolate) can cross the blood-brain barrier and is often used to treat some mitochondrial diseases and cerebral folate deficiency CFD and methylmalonic acid combined with homocystinemia. Folic acid is especially important for mitochondrial diseases associated with cerebral folate deficiency (such as polymerase gamma (POLG) deficiency, mtDNA) deletion, Kearns Sayre syndrome), which may cause white matter changes. This change may be reversible after supplementation with folinic acid (folinic acid), which is suitable for mitochondrial DNA depletion (including POLG) and Kearns Sayre syndrome.
Cerebral folate deficiency (CFD) causes autism, epilepsy, schizophrenia, and depression
1.3 Substrate supplementation and nitrogen removal agents
This type of therapy is common in the treatment of urea cycle disorders (UCD), which is a group of metabolic disorders that cause hyperammonemia. If patients are not treated in time, they will suffer from brain nerve damage such as intellectual and mental abnormalities.
Urea cycle substrates: arginine, citrulline
Nitrogen-removing agents: sodium benzoate, sodium phenylbutyrate, glyceryl phenylbutyrate oral solution
Guidelines for the diagnosis and management of urea cycle disorders and hyperammonemia
1.4 Liver transplantation
Inherited metabolic disorders (IEMs) are a significant cause of morbidity and mortality in children. Their clinical presentation is diverse and can include end-stage liver disease, hepatocellular carcinoma, renal tubular acidosis, epilepsy, encephalopathy, myopathy, and others. Liver transplantation (LT) is the treatment of choice for many metabolic diseases. LT is contraindicated for mitochondrial disease with significant extrahepatic involvement. Combined liver-kidney transplantation is indicated for disease in which the underlying defect severely impairs both organs. LT has excellent results in metabolic diseases. Adjuvant partial orthotopic LT is an attractive option as it delivers the defective enzyme, leaves the native liver intact, and has the option to remove immunosuppression in case gene therapy can be offered in the future.
Liver diseases caused by inherited metabolic disorders: the evolution of liver transplantation and the future prospects of hepatocyte transplantation
1.5 Future treatments for inherited metabolic disorders (IEMs)
The above therapies are the current treatments for IEM, none of which can cure this type of disease. In addition, some patients suffer from sequelae of brain damage due to failure to receive timely treatment. Because brain damage is irreversible, current therapies cannot cure brain damage. Future therapies will mainly focus on curative therapies: gene editing, enzyme replacement therapy, stem cell therapy, etc. Among them, gene editing and enzyme replacement therapy can cure this type of metabolic disorder in patients, and stem cell therapy can repair human nerves (brain nerves and muscle nerves, etc.). The combined treatment of these future therapies can change the current medical predicament of patients. Most of these therapies are currently in the research and development stage, and it is believed that they will be clinically used for patient treatment in the near future. At this stage, patients need to manage their diseases and nutrition and wait for the day of medical progress.
Future curative therapies for inherited metabolic disorders
2. Treatment of IEM disease and biological treatment of ASD
Combining the above introduction, we can find that there are many similarities between biological therapy for ASD and therapy for IEM diseases, because most genetic metabolic disorders will have manifestations of autism. After some autistic patients are diagnosed with IEM diseases in clinic, these patients can get timely treatment, and some patients' autism symptoms can be alleviated. Therefore, there are some hypotheses that IEM therapy can treat autism. However, there are thousands of IEMs, and there are many kinds of treatment methods involved. Some treatments are used in large doses beyond the instructions. Therefore, it is not recommended for ASD patients to try Shennong's tasting of herbs without being diagnosed with IEM disease.
Some ASD parents report that their children have made a lot of progress after receiving a certain type of biological therapy. In such cases, it is necessary to analyze whether the child is in a progressive stage of development. If the child does indeed benefit from progress and emotional stability through the use of a certain biological therapy, then it is necessary to analyze whether the benefits of these biological therapies mean that the patient has a metabolic disorder. For example, if the patient benefits from a low-protein diet, it is necessary to analyze whether the patient has amino acid or urea cycle disorders.
A patient had methylmalonic acid combined with homocysteine (an inherited metabolic disorder IEM) and showed symptoms of intellectual regression, autism, and mental disorders. He was treated with B12 injections and his abnormal neurological symptoms were alleviated. This does not mean that B12 can treat autism, but because this patient had methylmalonic acid combined with homocysteine and showed symptoms of autism, but B12 can treat methylmalonic acid combined with homocysteine, and the patient's clinical mental abnormalities were alleviated through correct treatment.
end
The treatment of IEM, an inherited metabolic disorder, has many similarities with the biological treatment of ASD. The treatment of IEM is to carry out treatment and monitoring under the guidance of a doctor after the disease is diagnosed. Through correct treatment, the disease of IEM patients is controlled and some abnormal mental symptoms are alleviated. ASD biological treatment is an experimental treatment for patients with autism/autism spectrum disorder ASD without a confirmed diagnosis. Because there is no monitoring by the medical management system, most treatments are private actions of ASD parents. This article systematically introduces the risks and benefits of biological treatments for ASD. For biological treatments that may be beneficial and have lower risks, it is recommended to be carried out under the guidance of doctors and nutritionists.
The current recommendations for people with autism/autism spectrum disorder (ASD) are:
The cause of ASD symptoms should be investigated based on the family situation, because at this stage a small number of ASD patients have been diagnosed with IEM genetic metabolic disorders. Through diagnosis and management of the disease, the abnormal manifestations of autism in these patients are alleviated, especially the core symptoms. Of course, the prognosis is related to whether the disease is treated early and in time. The earlier the diagnosis and treatment, the lighter the neurological sequelae, or even no neurological sequelae.
If the cause of ASD patients cannot be found, we can wait until IEM metabolomics research is more advanced in the future to find out the cause. For ASD patients who have undergone genetic testing, the genetic data can be saved and provided to other doctors for re-analysis in the future, or genetic companies can be asked to re-analyze them regularly.
At this stage, it is necessary to manage the nutritional management of ASD patients, as well as appropriate individual intervention training and suitable outdoor sports activities.
Nutritional management of inherited metabolic disorders
The important role of genetic testing in disease diagnosis, family inheritance pattern analysis and reproductive planning
Genetic testing and sequencing: whole genome (WGS), whole exome (WES), next-generation sequencing (NGS), mRNA sequencing (RNA-Seq)
Reference for this article: UPTODATE
0 notes
Text
Depression & Why I Tend to Isolate Myself...
First let me list a few things that I deal with daily and will probably never NOT deal with them… Just click the names below and you will get the meaning of what these illnesses are..
OCD
Posture Abnormalities
Eosinophilic Asthma
Anxiety
Major Depressive Disorder
Lagophthalmos Eye Disorder
Myasthenia Gravis
HYPOthyroidism
I was asked why do I shut myself off from the world. And even…

View On WordPress
0 notes
Text
I checked my blood test results on the patient portal. I'm pretty sure I'm anemic again. Iron binding capacity and transferrin levels are both high wile my iron saturation is low.
I also have high auto monocyte percent, eosinophil percent, and auto eosinophil absolute, along with LOW glucose and low carbon dioxide levels.
I looked each of these up:
The monocyte means that I could be fighting an infection, have an autoimmune disease, or a blood disorder.
High eosinophil may mean my body is fighting off an infection from a virus, bacteria, or fungus. So I'm likely also sick ATM.
Low c02 is low oxygen, which also could be a sign of other health issues.
Low glucose man's my blood sugar is also low.
And the iron and transferrin levels are indicating low iron and anemiaaaaaa
I love being medically unwell 24/7. 🫠
I see hematology on Thursday so hopefully these results get explained more. I was able to bump down to a lower deductible on m insurance that will go into affect Jun 1, so if I need major stuff again this year I'll pretty much hit my deductible immediately due to going from a $3500 threshold to $250.
My seasonal allergies have been kicking my ass this entire month, so that could b he reason why the blood work shows I'm potentially fighting an infection. My asthma acts up with said allergies and I always have to worry whether or not they turn into something worse like bronchitis or pneumonia due to the mucus buildup. I can't blow my nose due to extremely sensitive sinus cavities, so... I'm constantly sniffing and feel the mucus in the back of my throat. Hence why I worry about bronchitis and pneumonia.
Anyhow... this isn't so much of rant as it is an update I guess. Do I want to b anemic again? Hell no. But I KNOW that so.ething isn't right with my health and I need to figure out what it is.
I have a coworker who was clinically diagnosed with POTs and she even said that a lot of my symptoms sound like what she experienced for 6yrs leading up to her diagnosis. The only Hong I haven't had happen is a complete fainting spell, I have never passed out to my recollection. But doing research I know that not every POTs patient actually has episodes of syncope. There's just as many who only get pre-syncope and feel like passing out but never actually do.
I honestly hope I can get to the bottom of things soon.
1 note
·
View note
Text
When Blood Counts Matter: Indications for Differential Count Testing
The human body is a complex organism, and its intricate systems sometimes require thorough examination to understand their functioning fully. One such examination is the differential count test, a crucial component of a complete blood count (CBC). This test provides valuable insights into the composition of white blood cells (WBCs), aiding in the diagnosis and monitoring of various health conditions. Let's delve into the differential count test, its purpose, and who should consider taking it.
What is a Differential Count Test?
The Differential count test, also known as the white blood cell differential, is a laboratory analysis that identifies and quantifies the different types of white blood cells present in a blood sample. These cells include neutrophils, lymphocytes, monocytes, eosinophils, and basophils. Each type plays a distinct role in the immune system, and alterations in their levels can indicate specific health issues.
Purpose of the Test
1. Diagnosis of Infections:
Elevated levels of certain white blood cells, such as neutrophils, often indicate the presence of a bacterial infection. In contrast, increased lymphocytes may suggest a viral infection.
2. Immune System Disorders:
Abnormalities in white blood cell counts can signify underlying immune system disorders, such as leukemia, lymphoma, or autoimmune diseases.
3. Monitoring Treatment:
For individuals undergoing chemotherapy or other treatments affecting the bone marrow, regular differential count tests help monitor the effects on blood cell production.
4. Assessment of Allergic Reactions:
Eosinophils, in particular, are associated with allergic responses. Increased eosinophil counts may indicate allergies or parasitic infections.
5. Evaluation of Bone Marrow Function:
Changes in the distribution of white blood cell types can provide insights into bone marrow function and potential disorders affecting blood cell production.
Who Should Take It?
1. Routine Health Screenings:
Differential count tests are often included as part of routine health check-ups, especially for individuals with a history of immune system disorders or those undergoing treatment affecting blood cell counts.
2. Symptomatic Individuals:
People experiencing symptoms such as recurrent infections, unexplained fever, fatigue, or abnormal bruising may benefit from a differential count test to assess their immune system function.
3. Preoperative Evaluation:
Surgeons may request differential count tests before certain surgeries to ensure the patient's immune system is functioning optimally and to identify any underlying conditions that could affect surgery or recovery.
4. Monitoring Chronic Conditions:
Individuals with chronic illnesses, such as HIV/AIDS or autoimmune disorders, often require regular differential count tests to monitor disease progression and treatment effectiveness.
5. Cancer Patients:
Differential count tests are crucial for cancer patients undergoing treatment, as they help assess the impact of therapies on blood cell counts and detect any complications, such as infection or bone marrow suppression.
Conclusion:
The differential count test is a valuable tool in evaluating the composition of white blood cells and assessing immune system function. By identifying abnormalities in white blood cell counts, healthcare providers can diagnose infections, monitor treatment effectiveness, and detect underlying health conditions. Whether as part of routine screenings or in response to specific symptoms, individuals can benefit from undergoing this essential test to safeguard their health and well-being. Always consult with a healthcare professional to determine if a differential count test is appropriate for your situation.
0 notes